jvdesigns2002
Footballguy
I hope that you feel well soon GB.managed to avoid COVID all this time till Thursday.
after some temps, and sore throat, my biggest complaint is tight chest and a cough.
I hope that you feel well soon GB.managed to avoid COVID all this time till Thursday.
after some temps, and sore throat, my biggest complaint is tight chest and a cough.
Interesting. When my wife tested positive it lit up like right away and very dark.I think this sort of thing has been touched on upthread, but not really in much detail.
Now that the GOV is again giving out free rapid tests (http://covidtests.gov)If you do turn Positive this fall/winter, your rapid test may tell you more about your status (at the time of testing) than you think!How much time X how dark it gets
I wish we could embed tweets -- he's got a handy dandy little chart uploaded.

Covid Nasal Vaccines Get A Boost
Facts, data and analytics about biomedical matterserictopol.substack.com
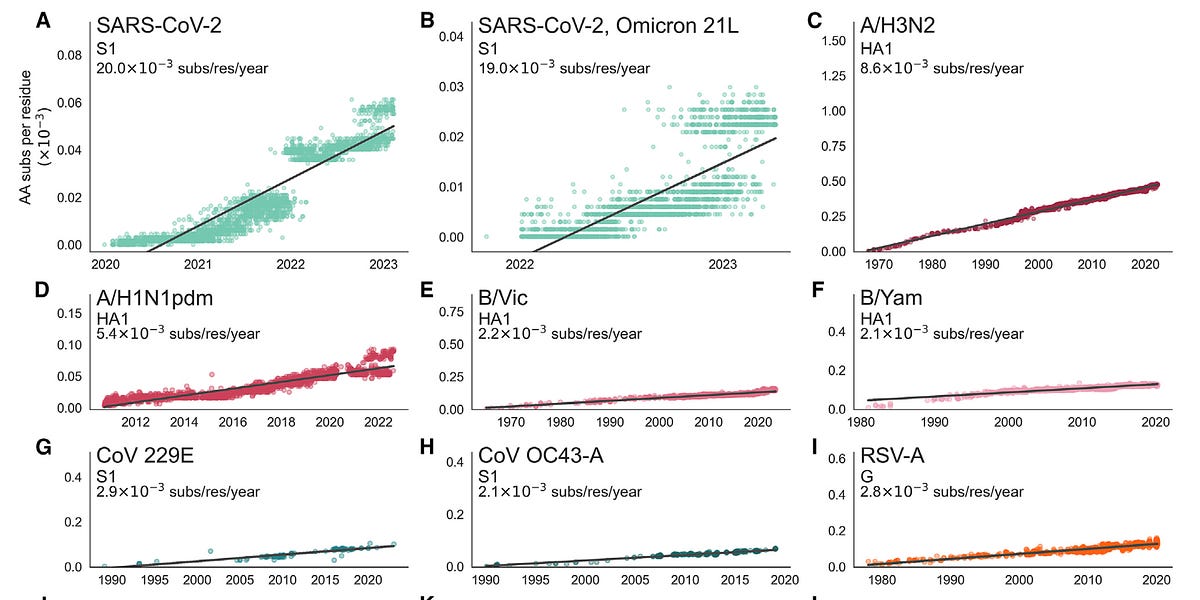
It looks like the last several attention-getting variants haven't really done much -- at least not in terms of increasing infirmity and affecting society. All varieties of COVID have spread so well, it doesn't seem like "it spreads more" has as much of an impact as one might expect any more. They all "spread more". It's always "spreading more".We won’t know for a few weeks as to whether JN.1 will be linked with a significant rise in Covid or how well our immune response from prior vaccinations, infection(s) and the XBB.1.5 new booster will keep us protected
Like the part in red. All COVID has always has "very high affinity" to the ACE2 receptor. So far as I understand it ... that's not a new feature of the virus.the lab data coming out for properties of BA.2.86 and particularly JN.1 are concerning. The Columbia University Ho lab report in Nature this week reinforced others for the very high affinity that BA.2.86 has towards the ACE2 receptor. which could lead to enhanced transmissibility.
Yes, that is true, but if my understanding is correct, there have been varying degrees of "stickiness" (my word) amongst the different variants. And this one is towards the "stickier" end of the scale.Like the part in red. All COVID has always has "very high affinity" to the ACE2 receptor. So far as I understand it ... that's not a new feature of the virus.the lab data coming out for properties of BA.2.86 and particularly JN.1 are concerning. The Columbia University Ho lab report in Nature this week reinforced others for the very high affinity that BA.2.86 has towards the ACE2 receptor. which could lead to enhanced transmissibility.
194 To corroborate these findings, we also evaluated the susceptibility of both BA.2.86 pseudoviruses
195 to neutralization by the dimeric human-ACE2-Fc protein, in comparison to BA.2, XBB.1.5, and
196 EG.5.1. In agreement with the SPR data, both versions of BA.2.86 were >2-fold more sensitive
197 to ACE2 inhibition than XBB.1.5 and EG.5.1, as determined by their IC50 values (Figure 4b). A
198 potential explanation for this heightened affinity may reside in the intrinsic charge properties of
199 the two interacting molecules. The region of human ACE2 targeted by the RBD is negatively
charged, while the Omicron RBD itself is positively charged32 200 . The higher receptor binding
201 affinity of the BA.2.86 spike might be attributed to the additional positive charges associated with
202 mutations V445H, N460K, N481K and A484K (Figure 4c). Only the N460K mutation is shared
203 with the spikes from XBB.1.5 and EG.5.1.253 Another scientific implication of our results is that the RBD of BA.2.86 is likely to be more
254 exposed than the RBD of XBB.1.5 or EG.5.1. This conclusion is inferred from the above
255 observation that the new variant is more sensitive than XBB.1.5 or EG.5.1 to neutralization by
256 class 1 and 4/1 mAbs, which target the “inner face” of RBD only when this domain is in the “up”
257 position. Since receptor binding also occurs when the RBD is “up”, this conclusion is in line
258 with the finding that the spike of BA.2.86 has a >2-fold higher affinity for the viral receptor
259 compared to the spike of XBB.1.5 or EG.5.1 (Figures 4a and 4b). In fact, BA.2.86 spike has
260 one of the highest receptor affinities we have measured, together with the spikes of some of the
viruses in the BA.2.75 sublineage
261 but the KD is undoubtedly determined by additional properties
262 including the electrostatic charge of the RBD (Figures 4c).
What?? You don't have random people just coming up and talking to you about the negative impacts of the vaccine?

Of course not. But they do tell me about their small weiners which I think is odd. https://www.totalwine.com/dynamic/490x/media/sys_master/twmmedia/hec/h39/26971202486302.pngWhat?? You don't have random people just coming up and talking to you about the negative impacts of the vaccine?
I know a little German ...Of course not. But they do tell me about their small weiners which I think is odd. https://www.totalwine.com/dynamic/490x/media/sys_master/twmmedia/hec/h39/26971202486302.pngWhat?? You don't have random people just coming up and talking to you about the negative impacts of the vaccine?
Saw this when you posted it. Sounds like it could also be a big breakthrough for thrombosis in general as well, regardless of cause.
Protein interaction causing rare but deadly vaccine-related clotting found - University of Birmingham
Discovery means potential protective treatments could be developed to block thrombosis from developing following vaccination.www.birmingham.ac.uk
I don't know about the fall 2023 (current) boosters, but I know that the 2021 Moderna shots were triple the dosage of the Pfizer ones. Some researchers felt like this caused more reports of side effects from the Moderna vaccine than the others.Just got my booster - first time getting the Pfizer one. The previous two were Moderna and made me feel like crap for about 12 hours approximately 12-18 hours after getting the shot. By that math, I'll feel like crap tomorrow morning and for most of the day. I have zero issue with the flu shot, but COVID has been awful since the 2nd dose of the 1st go-round.
My small glimmer of hope is that the tech who gave me the shot said she felt bad too with the Moderna version but felt a lot better with the Pfizer one, so maybe I'll be the same.
i got my 2nd ever flu shot and booster 2 weeks ago.
They made it pretty simple:
I got a text saying that I was eligible for the 2 shots with a link for pharmacy's around me to book online.
The pharmacy was 2 blocks away so went for lunch on the day of the appointment and it took 45 minutes out of my day.
My 1st flu shot was last year. The only reason I got it was because I was shopping at superstore and the announced free flu shots were available in 5 mins. I was 20 steps away, so had it done right away.
Never considered getting it before .
It is either my 2nd or 3rd booster since the 1st 2.
I had no side effects for any of the covid shots up to these 2 shots.i got my 2nd ever flu shot and booster 2 weeks ago.
They made it pretty simple:
I got a text saying that I was eligible for the 2 shots with a link for pharmacy's around me to book online.
The pharmacy was 2 blocks away so went for lunch on the day of the appointment and it took 45 minutes out of my day.
My 1st flu shot was last year. The only reason I got it was because I was shopping at superstore and the announced free flu shots were available in 5 mins. I was 20 steps away, so had it done right away.
Never considered getting it before .
It is either my 2nd or 3rd booster since the 1st 2.
How did you feel the next few days?
Scientists have discovered a gene variant, HLA-B*15:01, linked to asymptomatic COVID-19 cases, opening potential avenues for new treatments and vaccines.

My lady just got hers on Monday and she was down for 24 hours with headaches, chills and just feeling crappy.I don't know about the fall 2023 (current) boosters, but I know that the 2021 Moderna shots were triple the dosage of the Pfizer ones. Some researchers felt like this caused more reports of side effects from the Moderna vaccine than the others.Just got my booster - first time getting the Pfizer one. The previous two were Moderna and made me feel like crap for about 12 hours approximately 12-18 hours after getting the shot. By that math, I'll feel like crap tomorrow morning and for most of the day. I have zero issue with the flu shot, but COVID has been awful since the 2nd dose of the 1st go-round.
My small glimmer of hope is that the tech who gave me the shot said she felt bad too with the Moderna version but felt a lot better with the Pfizer one, so maybe I'll be the same.
I believe that Moderna halved the size of their doses for their 2022 bivalent boosters (still bigger than Pfizer, though). I'm sure it's discussed in this thread somewhere way back.
My lady just got hers on Monday and she was down for 24 hours with headaches, chills and just feeling crappy.I don't know about the fall 2023 (current) boosters, but I know that the 2021 Moderna shots were triple the dosage of the Pfizer ones. Some researchers felt like this caused more reports of side effects from the Moderna vaccine than the others.Just got my booster - first time getting the Pfizer one. The previous two were Moderna and made me feel like crap for about 12 hours approximately 12-18 hours after getting the shot. By that math, I'll feel like crap tomorrow morning and for most of the day. I have zero issue with the flu shot, but COVID has been awful since the 2nd dose of the 1st go-round.
My small glimmer of hope is that the tech who gave me the shot said she felt bad too with the Moderna version but felt a lot better with the Pfizer one, so maybe I'll be the same.
I believe that Moderna halved the size of their doses for their 2022 bivalent boosters (still bigger than Pfizer, though). I'm sure it's discussed in this thread somewhere way back.
Is that waning immunity we’re seeing in the most recent quarter?Fascinating chart here. Study looked at excess deaths by age and by quarter. Everything essentially turns instagreen when Omicron hit. COVID nerfed itself.
I think you also have to consider the cumulative immunity due to vaccination/prior infection. Restated, I don’t think the decrease in excess deaths is purely a function of a less deadly virus.Fascinating chart here. Study looked at excess deaths by age and by quarter. Everything essentially turns instagreen when Omicron hit. COVID nerfed itself.
The chart isn't from zero hedge. That's why I just included the picture.I think you also have to consider the cumulative immunity due to vaccination/prior infection. Restated, I don’t think the decrease in excess deaths is purely a function of a less deadly virus.Fascinating chart here. Study looked at excess deaths by age and by quarter. Everything essentially turns instagreen when Omicron hit. COVID nerfed itself.
Also of note, the highest excess mortality was seen in the 45-54 old age group - squarely in the FBG demographic.
Lastly, not sure I’d trust anything from zerohedge.
Is that waning immunity we’re seeing in the most recent quarter?Fascinating chart here. Study looked at excess deaths by age and by quarter. Everything essentially turns instagreen when Omicron hit. COVID nerfed itself.
Is that waning immunity we’re seeing in the most recent quarter?Fascinating chart here. Study looked at excess deaths by age and by quarter. Everything essentially turns instagreen when Omicron hit. COVID nerfed itself.
No -- that rightmost column is an aggregate percentage for the entire time period of the graphic -- April 2020 through June 2023.
...
Sand -- from where is that graph sourced? Can't tell by the link posted.

Bout that time again

Undiagnosed pneumonia outbreak in China puts pressure on pediatric hospitals, prompts questions
Media reports describe overwhelmed children's hospitals in multiple locations in China.www.cidrap.umn.edu
JUST IN—A new "mystery pneumonia" is tearing through China's schools. Hospitals are reportedly overwhelmed in Beijing and Liaoning with sick children and many classes are already cancelled.
Symptoms include inflammation in the lungs and a high fever but without a cough or other symptoms that normally come with respiratory illnesses.
The alert came Tuesday via ProMed, a global surveillance system that monitors outbreaks, and it was a ProMed in December of 2019 that first alerted the world to a virus outbreak in China that would later be called COVID.
Hey, pal don’t call me buddy!Bout that time again

Undiagnosed pneumonia outbreak in China puts pressure on pediatric hospitals, prompts questions
Media reports describe overwhelmed children's hospitals in multiple locations in China.www.cidrap.umn.edu
JUST IN—A new "mystery pneumonia" is tearing through China's schools. Hospitals are reportedly overwhelmed in Beijing and Liaoning with sick children and many classes are already cancelled.
Symptoms include inflammation in the lungs and a high fever but without a cough or other symptoms that normally come with respiratory illnesses.
The alert came Tuesday via ProMed, a global surveillance system that monitors outbreaks, and it was a ProMed in December of 2019 that first alerted the world to a virus outbreak in China that would later be called COVID.
Hey, screw you buddy and your thread!
Is that waning immunity we’re seeing in the most recent quarter?Fascinating chart here. Study looked at excess deaths by age and by quarter. Everything essentially turns instagreen when Omicron hit. COVID nerfed itself.
No -- that rightmost column is an aggregate percentage for the entire time period of the graphic -- April 2020 through June 2023.
...
Sand -- from where is that graph sourced? Can't tell by the link posted.

Might be here.Bout that time again

Undiagnosed pneumonia outbreak in China puts pressure on pediatric hospitals, prompts questions
Media reports describe overwhelmed children's hospitals in multiple locations in China.www.cidrap.umn.edu
JUST IN—A new "mystery pneumonia" is tearing through China's schools. Hospitals are reportedly overwhelmed in Beijing and Liaoning with sick children and many classes are already cancelled.
Symptoms include inflammation in the lungs and a high fever but without a cough or other symptoms that normally come with respiratory illnesses.
The alert came Tuesday via ProMed, a global surveillance system that monitors outbreaks, and it was a ProMed in December of 2019 that first alerted the world to a virus outbreak in China that would later be called COVID.
Pfft. Do you know how many people are trampled to death by hippos each year?Bout that time again

Undiagnosed pneumonia outbreak in China puts pressure on pediatric hospitals, prompts questions
Media reports describe overwhelmed children's hospitals in multiple locations in China.www.cidrap.umn.edu
JUST IN—A new "mystery pneumonia" is tearing through China's schools. Hospitals are reportedly overwhelmed in Beijing and Liaoning with sick children and many classes are already cancelled.
Symptoms include inflammation in the lungs and a high fever but without a cough or other symptoms that normally come with respiratory illnesses.
The alert came Tuesday via ProMed, a global surveillance system that monitors outbreaks, and it was a ProMed in December of 2019 that first alerted the world to a virus outbreak in China that would later be called COVID.
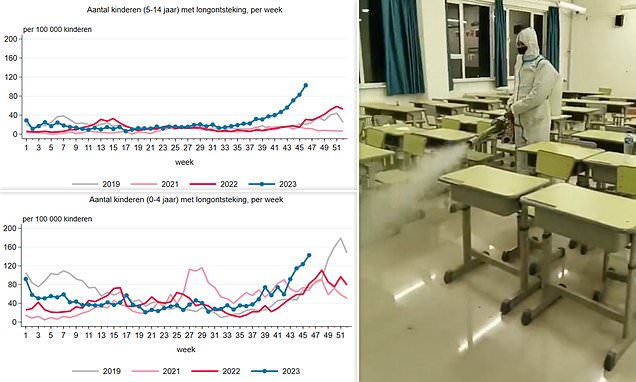


China brings back masks & social distancing over mystery outbreak
CHINA has brought back masks and social distancing in a chilling echo of lockdown as they battle a mystery pneumonia outbreak four years on from Covid. Alarming footage has emerged of mask-wearing …www.thesun.co.uk

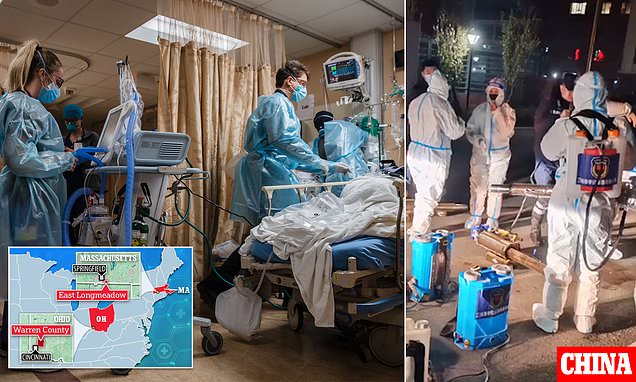
Nothing I've dug up thus far indicates that this is anything beyond normal respiratory season fodder. I think media hype mostly. The "white lung" increase does give me a little pause, but I don't think is a cause for concern other than monitoring.So this only affects kids? Do we know so little because it's China?

